|
|
|

|
|||||

|
|
Bristol Myers Squibb BMY has strengthened its near-term outlook following robust fourth-quarter 2025 results reported on Feb. 5, 2026. The stock rose 7.6% post-earnings, closing at $61.99 on Feb. 6 — near its 52-week high of $63.33 — reflecting improving investor sentiment.
Strength in BMY’s growth portfolio drove the company’s fourth-quarter earnings and revenue beat.
The going has been good for BMY in recent months. Shares of the company have surged 31.2% over the past three months, outperforming the industry’s growth of 11.9%. The stock has also outperformed the sector and the S&P 500 in this time frame.
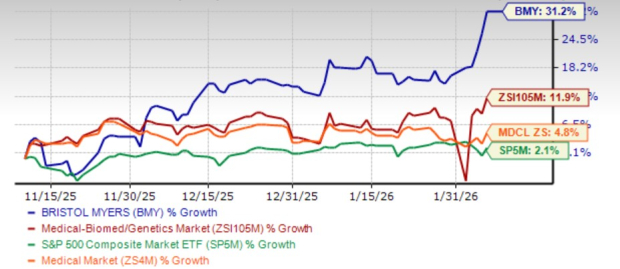
This sustained rally has helped restore confidence among previously cautious investors who now seem confident of the company’s ability to overcome generic erosion of legacy products.
Against this backdrop, let us examine BMY’s fundamentals to assess whether the stock makes for a prudent investment choice.
BMY’s Growth Portfolio includes key brands, such as Opdivo, Opdivo Qvantig, Orencia, Yervoy, Reblozyl, Camzyos, Breyanzi, Opdualag, Zeposia, Abecma, Sotyktu, Krazati and Cobenfy.
Revenues from this portfolio increased 16% to $7.4 billion in the fourth quarter, driven by the company’s immuno-oncology (IO) portfolio, along with drugs like Camzyos, Breyanzi and Reblozyl. The portfolio accounted for 59% of total revenues in the quarter.
Blockbuster IO drug Opdivo is the top revenue generator in this portfolio, generating $2.7 billion of sales in the fourth quarter, up 9% year over year. This was driven by label expansions in newer indications and continued share growth within the first-line non-small cell lung cancer setting.
The approval of Opdivo Qvantig (nivolumab and hyaluronidase-nvhy) for subcutaneous administration has further strengthened BMY’s IO franchise, with initial uptake proving robust across all approved tumor types in the United States.
Sales of its oncology drug, Opdualag, have also been robust, fueling the top line. Growth is strong in the US market, where the drug remains a standard of care in first-line melanoma.
Reblozyl, the thalassemia drug co-developed with Merck MRK, continues to be a major growth driver, with annualized sales now exceeding $2 billion. Revenue growth remains strong, reflecting solid uptake across first- and second-line MDS-associated anemia patients.
Breyanzi sales surged 49% in the fourth quarter. Yearly sales also surpassed a $1 billion annualized run rate, reflecting solid uptake in large B-cell lymphoma and contributions from additional indications. BMY is upbeat about Breyanzi’s prospects in 2026.
Cardiovascular drug Camzyos has also performed well, with revenues soaring 59% to $353 million on the back of robust demand.
The FDA approval of xanomeline and trospium chloride (formerly KarXT), marketed as Cobenfy, for the treatment of schizophrenia in adults, represents a significant milestone for BMY.
As the first novel pharmacological approach to schizophrenia in decades, Cobenfy has seen encouraging initial uptake, with sales of $155 million in 2025 driven by expanded access and deepened adoption across community and hospital settings.
The drug is expected to become a meaningful contributor to BMY’s revenue base over time, particularly as the company pursues label expansions into additional indications.
Collectively, these growth drivers are well positioned to sustain BMY’s top-line momentum in the coming quarters.
While BMY advances its growth portfolio, its legacy portfolio continues to face pressure from ongoing generic competition, particularly impacting Revlimid, Pomalyst, Sprycel and Abraxane. The erosion in sales from these mature brands has weighed on overall revenue performance.
Revenues from this portfolio fell 15% for the fourth quarter and full-year 2025.
BMY’s legacy portfolio also includes Eliquis, the blood thinner co-developed and co-commercialized globally with Pfizer PFE.
While demand increased for Eliquis, it was more than offset by expected continued generic impact across the remainder of the portfolio as well as the impacts from higher government channel rebates in the United States.
This portfolio accounted for 45% of total revenues of $48.2 billion in 2025. Management projects a decline of approximately 12-16% in the legacy portfolio in 2026, underscoring the magnitude of generic headwinds.
Nonetheless, Eliquis sales are expected to grow in the range of 10% to 15%, driven by continued global demand growth. The guidance includes the impact of the recent price reduction, which expands patient access and eliminates the associated inflation penalty.
We note that BMY recently announced a pricing agreement with the U.S. government under which it will supply Eliquis (apixaban) at no cost to Medicaid beginning Jan. 1, 2026. As part of the agreement, the company will also donate more than seven tons of the active pharmaceutical ingredient used in Eliquis to help strengthen supply-chain resilience.
Apart from Eliquis, the agreement also covers several other drugs — including Sotyktu, Zeposia, and Orencia SC (abatacept), among others — which will be offered at an 80% discount to cash-paying patients.
To further diversify its portfolio, Bristol Myers is making efforts to develop its pipeline. The company expects to report top-line registrational data (mostly in the second half) for six promising candidates — milvexian in both atrial fibrillation and secondary stroke prevention, admilparant in idiopathic pulmonary fibrosis, iberdomide, mezigdomide and arlo-cel in relapsed or refractory multiple myeloma and RYZ101 in second-line gastroenteropancreatic neuroendocrine tumors.
BMY also continues to pursue strategic acquisitions and collaborations to expand its pipeline. The recent acquisition of Orbital Therapeutics added OTX-201, Orbital’s lead preclinical RNA immunotherapy candidate currently in IND-enabling studies, to BMY’s pipeline. OTX-201 is a next-generation CAR T-cell therapy designed to reprogram cells in vivo, with the potential for a best-in-class profile in autoimmune diseases. The deal also adds Orbital’s proprietary RNA platform to BMY’s broader pipeline.
In 2025, BMY collaborated with BioNTech BNTX for the global co-development and co-commercialization of BioNTech’s investigational bispecific antibody pumitamig (BNT327) across numerous solid tumor types.
BMY and BioNTech announced positive first interim results from a global randomized phase II study evaluating pumitamig in combination with chemotherapy for patients with locally advanced or metastatic triple-negative breast cancer, regardless of PD-L1 expression.
The interim analysis demonstrated encouraging antitumor activity and a manageable safety profile for pumitamig plus chemotherapy in both first- and second-line treatment settings.
Bispecific antibodies that simultaneously target PD-1/PD-L1 and VEGF have emerged as a particularly attractive area in oncology. Pumitamig, a next-generation bispecific antibody candidate, is designed to target PD-L1 and VEGF-A, positioning it as a potentially differentiated therapy in cancer treatment.
Going by the price/earnings ratio, BMY is inexpensive as of now. Shares currently trade at 10.16x forward earnings, higher than its mean of 8.42x but lower than the large-cap pharma industry’s 18.76x.

The Zacks Consensus Estimate for 2026 EPS has moved north to $6.13 from $6.08 in the past seven days, while that for 2027 has inched up to $5.92 in the same time frame.

Bristol Myers Squibb delivered a resilient performance in 2025, supported by strong contributions from key growth drivers, such as Opdivo, Opdualag, Reblozyl, Breyanzi and Camzyos. These products helped stabilize the company’s revenue base despite ongoing generic erosion across its legacy portfolio.
Looking ahead, potential approvals of new drugs and label expansions for existing drugs should further diversify revenue streams. In addition, upcoming pipeline readouts represent meaningful near-term catalysts that could strengthen the long-term growth outlook of BMY’s growth portfolio.
On the profitability front, BMY is targeting $2 billion in annualized cost savings by the end of 2027. The company achieved approximately $1 billion in savings in 2025 and remains on track to realize the remaining savings through 2026-2027. As a result, operating expenses are expected to decline in 2026, supporting margin expansion.
Generic competition continues to weigh on the near-term revenue outlook. Management projects 2026 revenues of $46.0-$47.5 billion, down from $48.2 billion in 2025, reflecting continued pressure on legacy product sales.
We recommend prospective investors adopt a wait-and-watch approach for now and wait for more attractive entry levels.
For existing shareholders, holding the stock appears prudent, given the company’s attractive 4.07% dividend yield.
BMY currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
This article originally published on Zacks Investment Research (zacks.com).
| Jun-09 | |
| Jun-09 | |
| Jun-09 | |
| Jun-09 |
Pfizer's Hympavzi secures FDA expanded indications for haemophilia
PFE
Pharmaceutical Business Review
|
| Jun-09 | |
| Jun-09 | |
| Jun-08 | |
| Jun-08 | |
| Jun-08 | |
| Jun-08 | |
| Jun-08 | |
| Jun-08 | |
| Jun-08 | |
| Jun-08 | |
| Jun-08 |
Join thousands of traders who make more informed decisions with our premium features. Real-time quotes, advanced visualizations, alerts, and much more.
Learn more about Finviz Elite