|
|
|

|
|||||

|
|
Kymera KYMR reported a fourth-quarter 2025 loss of 97 cents per share, wider than the Zacks Consensus Estimate of a loss of 77 cents. In the year-ago quarter, Kymera reported a loss of 88 cents per share. The year-over-year deterioration was due to higher R&D expenses and lower collaboration revenues.
Collaboration revenues totaled $2.9 million, which missed the Zacks Consensus Estimate of $30 million. The revenues recognized in the reported quarter were attributable to the company’s collaboration with Gilead Sciences, Inc. GILD.
In the year-ago quarter, KYMR earned $7.4 million in collaboration revenues under its association with the pharma bigwig Sanofi SNY.
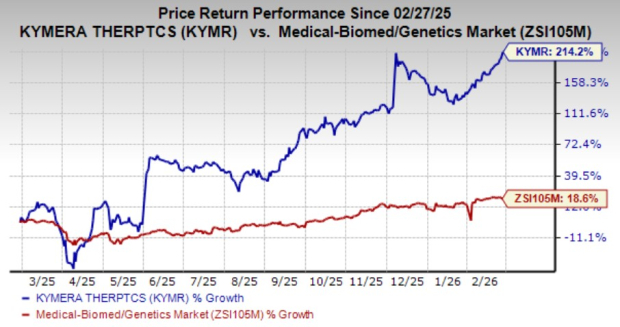
Shares of Kymera have rallied 214.2% in the past year compared with the industry’s rise of 18.6%.
Research and development expenses totaled $83.8 million, up 16.7% year over year. This was primarily due to increased expenses related to investments in the STAT6 program, platform, and discovery programs, as well as costs associated with continued growth in the research and development organization.
General and administrative expenses rose 3.7% year over year to $16.9 million, led by higher legal and professional service fees supporting growth, along with increased personnel, facility and related costs.
In December 2025, the company completed an underwritten equity offering that generated approximately $692 million in gross proceeds, including full exercise of the overallotment option. The financing increased year-end 2025 cash to about $1.6 billion and extended the cash runway into 2029.
Revenues decreased 16.8% to $39.2 million and missed the Zacks Consensus Estimate of $54.7 million.
Loss per share increased to $3.69 from $2.98 in 2024.
Kymera is using targeted protein degradation (TPD), a next-generation small molecule therapeutic modality that engages the body’s natural cellular recycling system to selectively eliminate disease-causing protein.
In its STAT6 Degrader Program, KT-621 is a first-in-class, once-daily oral STAT6 degrader in phase 2 development for atopic dermatitis (AD) and asthma, targeting the central driver of type II inflammation.
In December 2025, the company reported positive data from the phase Ib BroADen study on KT-621, which showed deep STAT6 degradation, significant biomarker reductions and meaningful clinical improvements across AD and related conditions, with favorable safety.
In January 2026, the BROADEN2 phase IIb AD study was expanded to include adolescents, while dosing began in the mid-stage BREADTH study in eosinophilic asthma. Both global, placebo-controlled studies are ongoing, with data expected from mid- to late-2027.
KT-621 also received Fast Track designation for moderate-to-severe AD in December 2025.
In the IRF5 Degrader Program, KT-579 is a first-in-class oral IRF5 degrader in phase 1 development, designed to suppress pro-inflammatory cytokines, Type I interferons and autoantibody production while preserving normal immune function.
Earlier this month, following IND clearance from the FDA, dosing began in a first-in-human phase I study in healthy volunteers to evaluate safety, pharmacokinetics, and pharmacodynamics, with data expected in the second half of 2026.
Among partnered programs, KT-485/SAR447971 is a selective, potent oral IRAK4 degrader being co-developed with Sanofi for immuno-inflammatory diseases. The program has completed IND-enabling studies and is expected to enter clinical trials in 2026.
Preclinical work is ongoing under an exclusive option and license agreement with GILD to advance the company’s oral CDK2 molecular glue program for breast cancer and other solid tumors. If the option is exercised, Gilead will pay an option fee and assume full responsibility for development, manufacturing and commercialization.
KYMR’s R&D expenses continue to increase as the company advances its pipeline.
The company remains well capitalized, ending 2025 with about $1.6 billion in cash and runway into 2029, supporting multiple mid- and early-stage programs. Kymera’s novel TPD strategy appears promising, and the pipeline progress is encouraging.
The investment thesis centers on pipeline execution in targeted protein degradation, led by KT-621 in phase 2 for atopic dermatitis and asthma. Additional positive pipeline updates on KT-621 will be a boost for the stock.
Kymera Therapeutics currently carries a Zacks Rank #3 (Hold). A better-ranked biotech is Castle Biosciences CSTL, which currently sports a Zacks Rank #1 (Strong Buy). You can see the complete list of today’s Zacks #1 Rank stocks here.
Over the past 60 days, Castle Biosciences’ 2026 loss per share estimates have narrowed from $1.06 to 96 cents. CSTL’s shares have rallied 26.2% over the past six months.
Castle Biosciences’ earnings beat estimates in three of the trailing four quarters and missed in the remaining one, with the average surprise being 66.11%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
This article originally published on Zacks Investment Research (zacks.com).
| 4 hours | |
| 4 hours | |
| 5 hours | |
| 14 hours | |
| 15 hours |
Sanofi and Owkin partner to develop next-generation biopharma AI agents
SNY
Pharmaceutical Business Review
|
| Jun-04 | |
| Jun-04 | |
| Jun-03 | |
| Jun-02 | |
| Jun-01 | |
| May-29 | |
| May-29 | |
| May-28 |
FDA accelerated approval of Hepcludex significant milestone for US HDV market
GILD
Clinical Trials Arena
|
| May-27 | |
| May-27 |
Join thousands of traders who make more informed decisions with our premium features. Real-time quotes, advanced visualizations, alerts, and much more.
Learn more about Finviz Elite