|
|
|

|
|||||

|
|
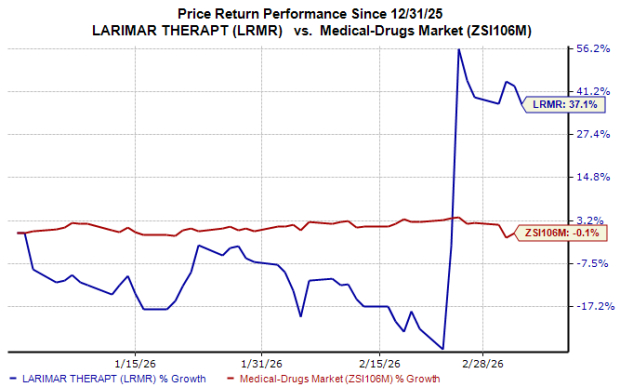
Shares of Larimar Therapeutics LRMR rose 55% in the past month after the company announced key regulatory updates related to nomlabofusp, its lead pipeline asset. Larimar outlined upcoming milestones related to the drug.
Last week, the company announced that the FDA granted breakthrough therapy designation (BTD) to nomlabofusp for the treatment of adults and children with Friedreich’s ataxia (FA), a rare neurodegenerative disorder caused by a mutation in the frataxin (FXN) gene.
The agency grants BTD to expedite the development and review of therapies for severe or life-threatening diseases where preliminary clinical evidence shows that therapy may provide substantial improvements over available treatments. The designation was granted after the FDA reviewed available clinical data from an ongoing open-label (OL) study evaluating nomlabofusp in adult and pediatric patients with FA.
Preliminary data from the OL study showed increases in skin FXN levels to those seen in asymptomatic carriers — people who carry the FA gene mutation but do not develop symptoms of the disease. The study showed consistent directional improvement across four key clinical outcomes after one year of treatment, including the modified Friedreich Ataxia Rating Scale (mFARS), FARS-Activities of Daily Living (ADL), the 9-Hole Peg Test (9-HPT) and the Modified Fatigue Impact Scale (MFIS).
These findings indicate that nomlabofusp may improve disease progression compared with the worsening typically observed in patients from the FA Clinical Outcome Measures Study (FACOMS), a long-running natural history study that monitors disease progression in untreated patients over time.
Alongside this update, Larimar said it received positive feedback from the FDA regarding a potential regulatory filing for nomlabofusp in FA. The agency indicated that it is willing to consider skin FXN levels as a novel surrogate endpoint to support a regulatory submission seeking accelerated approval for the therapy.
Top-line data from the ongoing OL study is expected in the second quarter of 2026. If the results are positive, Larimar plans to submit a regulatory filing to the FDA in June 2026. If approved, the company is targeting a commercial launch in the first half of next year.
The stock’s recent rally likely reflects improved investor confidence around the company’s pipeline, which is entirely dependent on nomlabofusp for growth. The BTD and alignment with the FDA on a surrogate endpoint suggest a potentially faster approval pathway for the drug. This is particularly meaningful because Larimar is a clinical-stage company with no marketed products, and nomlabofusp is the only drug in its pipeline. If approved, the therapy would become the company’s first commercial drug, marking a major milestone.
The opportunity is supported by a limited treatment landscape for FA. Currently, the only approved drug for this indication is Skyclarys, which is marketed by Biogen BIIB. The BIIB drug is the first FDA-approved therapy for the disease and has become a key revenue driver for the company.
Year to date, Larimar’s shares have risen 37% compared with the industry’s breakeven growth.
Nomlabofusp is an FXN protein replacement therapy designed to treat adults and children with FA.
Larimar announced plans to initiate a confirmatory late-stage study on the drug in the FA indication. It expects to dose the first patient in this study in mid-2026.
Larimar currently carries a Zacks Rank #3 (Hold).

Larimar Therapeutics, Inc. price | Larimar Therapeutics, Inc. Quote
Some better-ranked stocks from the sector are ANI Pharmaceuticals ANIP and ALX Oncology Holdings ALXO, each carrying a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Over the past 60 days, estimates for ANI Pharmaceuticals’ 2026 EPS have increased from $8.20 to $8.38, while the same for 2027 have risen from $9.25 to $9.90. The stock has declined 4% year to date.
ANI Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 22.21%.
Over the past 60 days, ALX Oncology’s 2026 loss per share estimates have narrowed from $1.21 to 88 cents, while the same for 2027 have narrowed from 99 cents to 60 cents. ALXO shares have rallied over 89% year to date.
ALX Oncology’s earnings missed estimates in each of the trailing four quarters, with the average negative surprise being 12.82%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
This article originally published on Zacks Investment Research (zacks.com).
| May-01 | |
| Apr-30 | |
| Apr-30 | |
| Apr-30 | |
| Apr-30 | |
| Apr-29 | |
| Apr-29 | |
| Apr-29 | |
| Apr-29 | |
| Apr-29 |
Biogen Walloped Wall Street's First Quarter Views; Here's What Happened
BIIB +6.00%
Investor's Business Daily
|
| Apr-27 | |
| Apr-24 |
Biogen seeks Darzalex rivalry in China for multiple myeloma with felzartamab deal
BIIB
Pharmaceutical Technology
|
| Apr-24 | |
| Apr-22 | |
| Apr-22 |
Join thousands of traders who make more informed decisions with our premium features. Real-time quotes, advanced visualizations, alerts, and much more.
Learn more about Finviz Elite