|
|
|

|
|||||

|
|
New Elite Feature: Compare relative returns, fundamentals, and sector rankings head-to-head.
AngioDynamics ANGO recently announced expanded indications in Europe for its NanoKnife System, broadening its clinical utility in soft tissue tumor ablation. The expanded approval covers NanoKnife’s use for soft tissue ablation for tumors of the liver, pancreas, kidney and prostate, including intermediate-risk prostate cancer cases.
The broadened indications expand physician access to the NanoKnife System’s irreversible electroporation (IRE) technology across multiple high-value oncology applications, enhancing its utility in treating tumors that are challenging to remove or situated near vital anatomical structures.
Management noted that the NanoKnife prostate indication continues to see increasing clinical adoption, with the company focused on driving further global expansion. The recent European approval of additional organ indications highlights the broader clinical applicability of its IRE technology and reinforces NanoKnife’s positioning as a scalable platform for complex tumor ablation. The company aims to broaden physician access to differentiated technologies that meaningfully expand treatment options across multiple disease areas.
To support broader clinical adoption and longitudinal evidence generation, ANGO plans to launch the LIVER-IRE Global Registry in collaboration with Ajith Siriwardena at the University of Manchester. The prospective registry will assess real-world outcomes in patients treated with IRE for liver tumors, further expanding the clinical evidence base for multi-organ applications of the NanoKnife System.
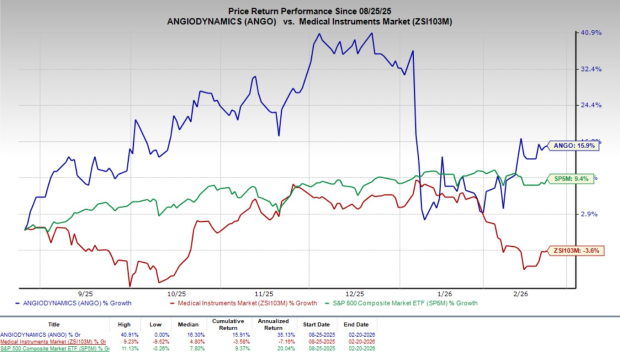
Shares of ANGO have gained 0.2% since the announcement on Thursday. Over the past six months, shares of the company have climbed 15.9% against the industry’s 3.6% decline. However, the S&P 500 has risen 9.4% during the same time frame.
In the long run, ANGO is positioned for significant growth following the European expansion of indications for its NanoKnife System. Broader approvals across liver, pancreas, kidney and prostate tumors significantly expand its addressable market and strengthen its multi-organ oncology platform. With Europe representing a sizable share of global IRE procedures and volumes continuing to rise, the company stands to benefit from higher physician adoption and recurring procedure growth. The launch of the LIVER-IRE Global Registry further reinforces clinical credibility, supporting long-term demand, competitive differentiation and revenue expansion across key international markets.
ANGO currently has a market capitalization of $462.46 million.
Unlike conventional radiofrequency or microwave ablation technologies, the NanoKnife System leverages IRE, a non-thermal ablation modality that destroys tumor cells while preserving the structural integrity of blood vessels, bile ducts and nerves. This differentiated mechanism enhances its clinical utility in treating tumors situated within anatomically complex regions like the pancreas and liver.
Clinical evidence for the NanoKnife System’s IRE platform continues to build across multiple solid tumor indications. Data from prospective and multicenter trials indicate procedural success and promising clinical outcomes in metastatic colorectal cancer, as well as in liver, pancreatic, hepatocellular and renal malignancies. Comparative studies and real-world evidence have reinforced the technology’s safety and value in managing tumors located in sensitive regions.
Europe contributes about 28% of total global IRE procedure volumes, underscoring its significance within the overall addressable market. Annual worldwide IRE procedures are estimated to have exceeded 45,000, supported by increasing cancer prevalence, rising clinician adoption, continued improvements in image-guided delivery and sustained preference for minimally invasive treatment modalities.
Against this backdrop, the expanded indications enhance the NanoKnife System’s ability to capture incremental share within the European market and further solidify its positioning as a versatile, multi-organ tumor ablation platform.
Going by the data provided by Precedence Research, the tumor ablation market is valued at $2.47 billion in 2026 and is expected to witness a CAGR of 13.1% through 2035.
Factors like the rising demand for minimally and non-invasive therapies, the availability of advanced therapeutic options, the integration of image-guided techniques and the emergence of novel ablation methods are boosting the market’s growth.
AngioDynamics, in collaboration with The PERT Consortium, recently announced the launch of the ALPHA-PE Research Fund, an investigator-initiated program supporting independent research in pulmonary embolism (PE). PE impacts 1 in 1,000 individuals annually and is associated with over 50,000 deaths per year in the United States, highlighting the need for continued clinical innovation. The ALPHA-PE Research Fund aims to address evidence gaps in PE management by funding physician-led, real-world studies to generate clinical data and inform treatment strategies.
During the second quarter, the FDA approved ANGO’s IDE application for the APEX-Return pivotal trial evaluating the AlphaReturn Blood Management System in combination with the AlphaVac F1885 MMA System for acute PE.
The FDA also approved the IDE for the PAVE (Percutaneous AngioVac Vegetation Extraction) pilot study, a prospective, single-arm, multicenter feasibility trial enrolling up to 30 patients across six U.S. sites. The study will evaluate the AngioVac System for the percutaneous removal of right heart vegetation in patients with right-sided infective endocarditis, targeting a high-risk population with limited treatment options.
ANGO received 510(k) clearance for a modified AlphaVac F1885 System with expanded indications, allowing aspiration and contrast/fluid injection. The updated sheath facilitates endovascular device insertion while minimizing blood loss.

AngioDynamics, Inc. price | AngioDynamics, Inc. Quote
Currently, ANGO flaunts Zacks Rank #1 (Strong Buy).
Some other top-ranked stocks from the broader medical space are Intuitive Surgical ISRG, Veracyte VCYT and Cardinal Health CAH.
Intuitive Surgical, sporting a Zacks Rank #1 at present, reported fourth-quarter 2025 adjusted earnings per share (EPS) of $2.53, beating the Zacks Consensus Estimate by 12.4%. Revenues of $2.87 billion surpassed the Zacks Consensus Estimate by 4.7%. You can see the complete list of today’s Zacks #1 Rank stocks here.
ISRG has an estimated long-term earnings growth rate of 15.7% compared with the industry’s 13% rise. The company beat earnings estimates in the trailing four quarters, the average surprise being 13.2%.
Veracyte, currently carrying a Zacks Rank #2 (Buy), reported a third-quarter 2025 adjusted EPS of 51 cents, which surpassed the Zacks Consensus Estimate by 59.4%. Revenues of $131.8 million beat the Zacks Consensus Estimate by 5.5%.
VCYT has an estimated earnings recession rate of 3% for 2026 compared with the industry’s 17.4% rise. The company beat earnings estimates in the trailing four quarters, the average surprise being 45.1%.
Cardinal Health, currently carrying a Zacks Rank #2, reported a second-quarter fiscal 2026 adjusted EPS of $2.63, which surpassed the Zacks Consensus Estimate by 10%. Revenues of $65.6 billion beat the Zacks Consensus Estimate by 0.9%.
CAH has an estimated long-term earnings growth rate of 15% compared with the industry’s 9.4% rise. The company beat earnings estimates in the trailing four quarters, the average surprise being 9.3%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
This article originally published on Zacks Investment Research (zacks.com).
| 5 hours | |
| May-30 | |
| May-29 | |
| May-28 | |
| May-28 | |
| May-28 | |
| May-27 | |
| May-22 | |
| May-21 | |
| May-21 | |
| May-19 | |
| May-19 | |
| May-15 | |
| May-13 | |
| May-11 |
Join thousands of traders who make more informed decisions with our premium features. Real-time quotes, advanced visualizations, alerts, and much more.
Learn more about Finviz Elite