|
|
|

|
|||||

|
|
Recursion Pharmaceuticals RXRX reported a loss of 21 cents per share in the fourth quarter of 2025, narrower than the Zacks Consensus Estimate of a loss of 28 cents. The company had incurred a loss of 53 cents per share in the year-ago quarter.
In the absence of an approved product, Recursion Pharmaceuticals only recognizes collaboration and grant revenues from its partners. Total revenues for the quarter were $35.5 million, which rose sharply year over year, largely driven by a $30 million milestone payment from Roche for the second phenomap in October 2025, of which a portion was recognized in the fourth quarter. Revenues also benefited from higher contributions tied to the company’s collaboration with Sanofi. The reported figure beat the Zacks Consensus Estimate of $26 million.
RXRX also recognizes periodic revenues from its ongoing collaboration agreements with Bayer and Merck KGaA, Darmstadt, Germany. Shares of the company are gaining in the pre-market hours on the back of better-than-expected fourth-quarter results.
In the fourth quarter of 2025, Research and development (R&D) expenses decreased 2% to $95.9 million. The downtick in R&D expenses can be attributed to improved operating efficiency and a strategic reprioritization of the clinical portfolio in the second quarter of 2025.
General and administrative (G&A) expenses were $33.7 million in the reported quarter, down 56% year over year, primarily due to the inclusion of transaction expenses from the business combination with Exscientia in the year-ago quarter. Additionally, Recursion Pharmaceuticals’ cost of revenues in the reported quarter increased 12% to $14.3 million.
The company had cash, cash equivalents and restricted cash worth $753.9 million as of Dec. 31, 2025, compared with $667.1 million as of Sept. 30, 2025. Recursion Pharmaceuticals expects its existing cash, cash equivalents and restricted cash to fuel operations into early 2028, based on its current business plan.
RXRX shares have plunged 26.9% in the past six months against the industry’s 23% growth.
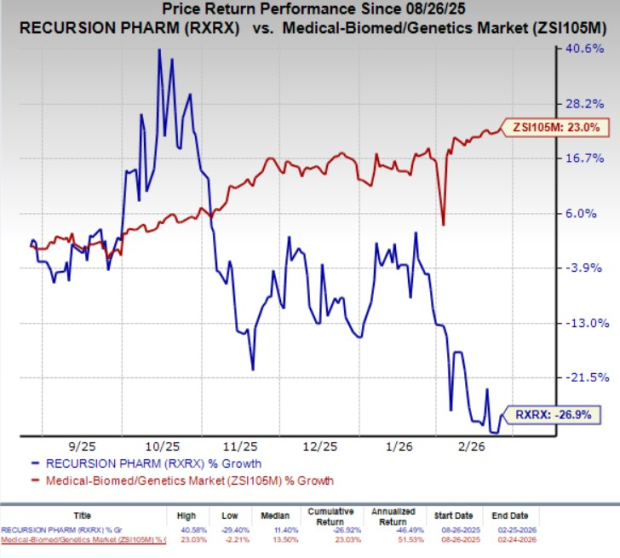
In 2025, Recursion Pharmaceuticals recorded total revenues of $74.7 million, which increased 27% year over year and beat the Zacks Consensus Estimate of $64.6 million.
The company reported a loss per share of $1.44 in 2025, narrower than the loss of $1.69 per share incurred in 2024. The reported figure was also narrower than the Zacks Consensus Estimate of a loss of $1.59 per share.
Following a strategic reprioritization in 2025, Recursion Pharmaceuticals has shifted its focus and resources to the development of other candidates in its clinical pipeline. Such candidates include REC-4881, which is being developed for familial adenomatous polyposis in a phase Ib/II TUPELO study. In May 2025, the company reported preliminary data from this study and reported additional positive data in December 2025. REC-4881 drove rapid and durable reductions in polyp burden in FAP patients, providing the first clinical validation of the Recursion OS platform. The company plans to engage with the FDA in the first half of 2026 to discuss a potential registration pathway, while also expanding eligibility to younger patients and further refining the dosing strategy.
In 2024, Recursion Pharmaceuticals initiated its phase I/II DAHLIA study of REC-1245, a new chemical entity for the treatment of biomarker-enriched solid tumors and lymphoma. Data readout from the phase I dose-escalation portion of the DAHLIA study is expected in the first half of 2026. Recursion Pharmaceuticals is also developing a few other candidates, like REC-617 (advanced solid tumors) and REC-3565 (B-cell malignancies), in separate early-stage studies.
In 2025, Recursion Pharmaceuticals acquired Rallybio’s full stake in their joint venture for developing REV102 (now REC102) and an associated backup molecule for the treatment of hypophosphatasia, a rare and debilitating genetic disorder. REC102, a potent and selective ENPP1 inhibitor with strong preclinical safety data, is expected to enter phase I studies by late 2026. Its oral formulation offers a major advantage over current enzyme replacement therapies, potentially improving patient adherence and reducing treatment-associated risks.

Recursion Pharmaceuticals, Inc. price-consensus-eps-surprise-chart | Recursion Pharmaceuticals, Inc. Quote
Recursion Pharmaceuticals currently carries a Zacks Rank #3 (Hold).
Some better-ranked stocks in the biotech sector are Harmony Biosciences HRMY, Immunocore IMCR and Castle Biosciences CSTL, each sporting a Zacks Rank #1 (Strong Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.
Over the past 60 days, estimates for Harmony Biosciences’ 2026 earnings per share have risen from $3.72 to $4.00. HRMY shares have lost 24.1% over the past six months.
Harmony Biosciences’ earnings beat estimates in two of the trailing four quarters, missing the mark on the other two occasions, with the average surprise being 7.20%.
Over the past 60 days, estimates for Immunocore’s 2026 loss per share have narrowed from 97 cents to 90 cents. IMCR shares have risen 3.3% over the past six months.
Immunocore’s earnings beat estimates in three of the trailing four quarters and missed in the remaining one, with the average earnings surprise being 53.96%.
Over the past 60 days, estimates for Castle Biosciences’ 2026 loss per share have narrowed from $1.06 to 96 cents. CSTL shares have surged 40.5% over the past six months.
Castle Biosciences’ earnings beat estimates in three of the trailing four quarters and missed in the remaining one, with the average surprise being 66.11%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
This article originally published on Zacks Investment Research (zacks.com).
| Jun-04 | |
| Jun-02 | |
| May-31 | |
| May-29 | |
| May-27 | |
| May-27 | |
| May-13 | |
| May-12 | |
| May-07 | |
| May-06 | |
| May-06 | |
| May-06 | |
| May-06 | |
| May-06 | |
| May-06 |
Join thousands of traders who make more informed decisions with our premium features. Real-time quotes, advanced visualizations, alerts, and much more.
Learn more about Finviz Elite