|
|
|

|
|||||

|
|
Iovance Biotherapeutics IOVA incurred a fourth-quarter 2025 loss of 18 cents per share, narrower than the Zacks Consensus Estimate of a loss of 22 cents. In the year-ago quarter, the company reported a loss per share of 26 cents.
Total revenues for the fourth quarter rose 17.6% year over year to $87 million, generated entirely from the sales of the company’s two marketed drugs. The top line beat the Zacks Consensus Estimate of $75 million.
Iovance currently has two marketed drugs in its portfolio — the IL-2 product Proleukin and the TIL therapy Amtagvi. While Proleukin is approved to treat metastatic renal cell carcinoma and metastatic melanoma in adults, Amtagvi is approved for the advanced melanoma indication.
IOVA recorded approximately $65 million from Amtagvi sales during the quarter, representing a 33.4% year-over-year increase, driven by robust demand. This figure beat the Zacks Consensus Estimate of $61 million.
Proleukin generated $22 million during the quarter, down 12% year over year. The figure beat the Zacks Consensus Estimate of $15 million.
Research & development expenses totaled $71.2 million in the fourth quarter, up 0.3% from the year-ago period.
Selling, general and administrative expenses declined 14% from the prior-year quarter’s figure to $36.4 million, mainly due to lower stock compensation expenses.
As a result of a restructuring plan initiated in August, Iovance has started experiencing the benefits of cost optimization. The company reported a gross margin of 50% in the fourth quarter compared with 43% in the previous quarter, driven by improved operational efficiency.
As of Dec. 31, 2025, Iovance had cash, cash equivalents and investments of $303 million compared with $307 million as of Sept. 30, 2025. Management now expects its existing cash balance to fund operations into the third quarter of 2027.
For 2025, Iovance reported total revenues of approximately $264 million, representing a 61% year-over-year increase. Revenues were within the guidance range of $250 million to $300 million in the first full year of Amtagvi’s launch.
The company recorded a net loss per share of $1.09, narrower than a loss of $1.28 per share in 2024.
In a separate press release, the company reported encouraging data from an early-stage study evaluating Amtagvi in heavily pretreated advanced undifferentiated pleomorphic sarcoma (UPS) and dedifferentiated liposarcoma (DDLPS) patients. Data from the study showed that patients treated with the therapy achieved an objective response rate (ORR) of 50%.
Based on this positive data, Iovance plans to start a single-arm registrational study in second-line advanced UPS and DDLPS in the second quarter of 2026. It also intends to engage with the FDA to discuss a potential regulatory pathway for approval.
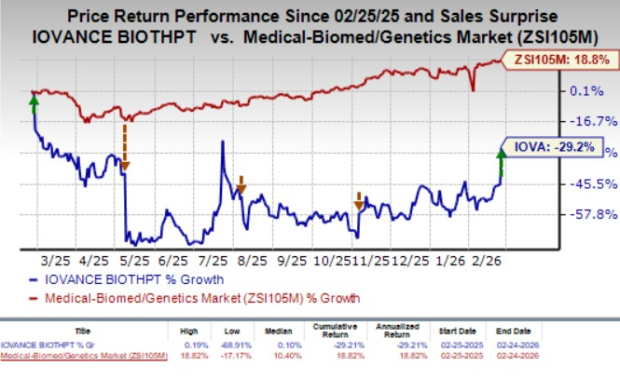
Shares of IOVA rose 31% on Tuesday after the company announced the above results. Investors cheered this positive update as treatment options for advanced UPS and DDLPS are limited. Recent studies have reported ORRs of less than 5%. The company's better-than-expected fourth-quarter results and growth in Amtagvi sales also impressed investors. However, over the past year, the stock has declined 29.2% against the industry’s 18.8% growth.
Regulatory applications for Amtagvi in the melanoma indication are under review, with potential approvals in the United Kingdom and Australia in the first half of 2026. In 2025, IOVA voluntarily withdrew its regulatory filing in the European Union due to a lack of alignment with the European Medicines Agency on the clinical data supporting the submission. The company is in discussions with the agency to resubmit a marketing authorization application in 2026.
Iovance continues to advance its development programs for Amtagvi. It is evaluating the drug in combination with Merck’s Keytruda in the phase III TILVANCE-301 study as a potential treatment for frontline advanced melanoma. This study will also serve as a confirmatory study seeking full approval for Amtagvi in the melanoma indication.
Beyond melanoma, Iovance is developing Amtagvi in patients with previously treated advanced non-squamous non-small cell lung cancer (NSCLC) under the phase II IOV-LUN-202 study. In November 2025, IOVA provided an interim update from the IOV-LUN-202 study, which showed that treatment with Amtagvi achieved an objective response rate of around 26%, which is twice the number compared to the current standard of care chemotherapy.
Based on these results, the company intends to submit a supplemental biologics license application to the FDA seeking approval for expanded use in the NSCLC indication later this year, with a potential launch in the second half of 2027.
Amtagvi is being evaluated in separate mid-stage studies for previously treated advanced endometrial cancer and melanoma.

Iovance Biotherapeutics, Inc. price-consensus-eps-surprise-chart | Iovance Biotherapeutics, Inc. Quote
Iovance currently has a Zacks Rank #3 (Hold).
Some better-ranked stocks in the biotech sector are Harmony Biosciences HRMY, Assertio Holdings ASRT and Castle Biosciences CSTL, each currently sporting a Zacks Rank #1 (Strong Buy). You can see the complete list of today’s Zacks #1 Rank stocks here.
Over the past 60 days, estimates for Harmony Biosciences’ 2026 earnings per share have risen from $3.72 to $4.00. HRMY shares have lost 23.7% over the past year.
Harmony Biosciences’ earnings beat estimates in two of the trailing four quarters but missed in the remaining quarters, with the average surprise being 7.20%.
Over the past 60 days, estimates for Assertio’s 2026 loss per share have narrowed from 30 cents to 28 cents. ASRT shares have gained 1.6% over the past year.
Assertio’s earnings beat estimates in one of the trailing four quarters and missed in the remaining three quarters, with the average negative surprise being 35.21%.
Over the past 60 days, estimates for Castle Biosciences’ 2026 loss per share have narrowed from $1.06 to 96 cents. CSTL shares have risen 19.5% over the past year.
Castle Biosciences’ earnings beat estimates in three of the trailing four quarters and missed in the remaining one, with the average surprise being 66.11%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
This article originally published on Zacks Investment Research (zacks.com).
| Jun-04 | |
| Jun-03 | |
| Jun-01 | |
| May-27 | |
| May-25 | |
| May-22 | |
| May-21 | |
| May-19 | |
| May-13 | |
| May-13 | |
| May-13 | |
| May-12 | |
| May-08 | |
| May-07 | |
| May-07 |
Join thousands of traders who make more informed decisions with our premium features. Real-time quotes, advanced visualizations, alerts, and much more.
Learn more about Finviz Elite