|
|
|

|
|||||

|
|
Acadia Pharmaceuticals ACAD reported fourth-quarter 2025 earnings per share (EPS) of 16 cents (excluding tax benefit), which beat the Zacks Consensus Estimate of 12 cents. In the year-ago quarter, the company had reported adjusted EPS of 17 cents.
Including the tax benefit recognized from a one-time impact of the One Big Beautiful Bill Act, the company reported EPS of $1.60 for the fourth quarter of 2025 compared with EPS of 86 cents in the year-ago quarter.
In the fourth quarter, Acadia recorded total revenues of $284 million, which missed the Zacks Consensus Estimate of $293 million. ACAD’s net product revenues comprise sales of its two marketed products, Nuplazid (pimavanserin) and Daybue (trofinetide).
Acadia’s first drug, Nuplazid, is approved in the United States for the treatment of hallucinations and delusions associated with Parkinson’s disease psychosis. ACAD’s second product, Daybue, received approval in 2023 for treating Rett syndrome in adult and pediatric patients aged two years and older. The drug was launched in the United States in April 2023.
Total revenues increased 9% year over year, driven by contributions from Daybue and continued growth in Nuplazid's market share.
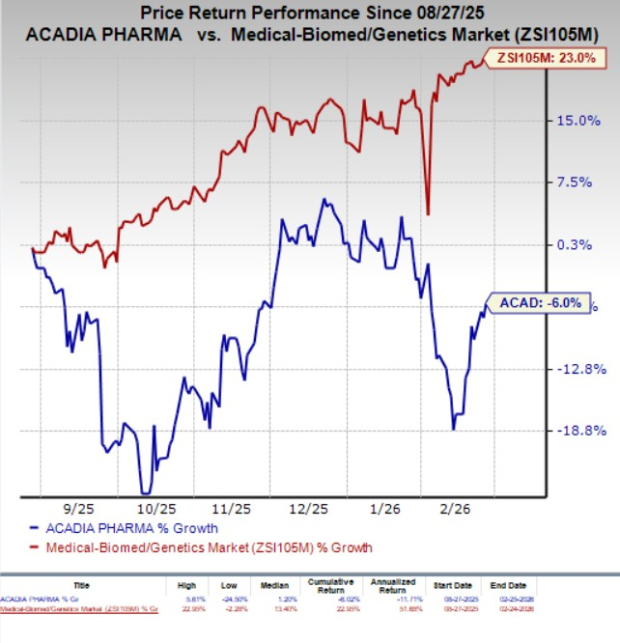
In the past six months, shares of Acadia have lost 6% against the industry’s 23% growth.
Revenues from Nuplazid increased 7% year over year to $174.4 million in the fourth quarter of 2025, driven primarily by volume growth. Nuplazid sales missed the Zacks Consensus Estimate of $184.1 million.
Daybue recorded net product sales of $109.6 million in the reported quarter, up 13% year over year, driven by the growth in the drug’s unit sales as Acadia shipped to more unique patients. The reported figure was a tad higher than the Zacks Consensus Estimate of $108.4 million.
Research and development (R&D) expenses were $84.8 million, down 16% year over year, due to a $28 million upfront business development payment for ACP-711 that was made in the year-ago quarter to Saniona.
Selling, general and administrative (SG&A) expenses were $155.6 million, up 20% year over year, owing to increased marketing investments to support Nuplazid and Daybue field expansion and marketing costs.
Acadia had cash, cash equivalents and investments worth $820 million as of Dec. 31, 2025, compared with $847 million as of Sept. 30, 2025.
In 2025, Acadia recorded total revenues of $1.07 billion, representing 12% year-over-year growth. The figure, however, missed the Zacks Consensus Estimate of $1.08 billion.
The company reported adjusted EPS of 82 cents in 2025, which beat the Zacks Consensus Estimate of 80 cents. Acadia reported GAAP EPS of $2.30 in 2025 compared with $1.36 reported in 2024.
Acadia expects total revenues from the U.S. sales of its products to be in the range of $1.22-$1.28 billion in 2026. Nuplazid net product sales are expected to be in the range of $760-$790 million, while U.S. sales of Daybue are expected to be between $460 million and $490 million.
R&D expenses in 2026 are projected to be in the range of $385-$410 million, while SG&A expenses are expected to be between $660 million and $700 million.

ACADIA Pharmaceuticals Inc. price-consensus-eps-surprise-chart | ACADIA Pharmaceuticals Inc. Quote
A regulatory filing for trofinetide to treat Rett syndrome in adults and pediatric patients aged two years and above is currently under review in the EU.
However, earlier this month, Acadia announced that it expects to receive a negative opinion from the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) for the marketing application for trofinetide to treat Rett syndrome, following a recent oral explanation.
Acadia was informed by the CHMP of a negative trend vote on its marketing application for trofinetide to treat Rett syndrome. Pending the outcome of the CHMP vote this month, Acadia plans to seek a re-examination of the CHMP opinion once it is formally adopted. With the CHMP likely to reject the marketing application for trofinetide in Rett syndrome, a potential approval in the EU is likely to be delayed.
During the quarter, the FDA approved Daybue Stix (trofinetide) for oral solution, a dye- and preservative-free powder formulation for the treatment of Rett syndrome in adults and pediatric patients aged two years and older. The new product expands the Daybue franchise, which remains the only FDA-approved treatment option for this indication. Acadia plans a limited launch of Daybue Stix during the first quarter of 2026, followed by broader availability in the second quarter. The company will continue to offer the current oral solution alongside the new formulation, strengthening its positioning in the Rett syndrome treatment market.
Acadia also anticipates a data readout from the phase II RADIANT study of ACP-204 (remlifanserin) for Alzheimer’s disease psychosis, which is on track for the August to October 2026 timeframe, representing a potential catalyst for the company this year.
Acadia currently carries a Zacks Rank #3 (Hold).
Some better-ranked stocks in the biotech sector are USANA Health Sciences USNA, Castle Biosciences CSTL and Recursion Pharmaceuticals RXRX. While USNA and CSTL sport a Zacks Rank #1 (Strong Buy) each, RXRX has a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.
Over the past 60 days, estimates for USANA Health Sciences’ 2026 EPS have risen from $1.90 to $2.00. USNA shares have lost 33.8% over the past six months.
USANA Health Sciences’ earnings beat estimates in three of the trailing four quarters and matched once, with the average surprise being 21.92%.
Over the past 60 days, estimates for Castle Biosciences’ 2026 loss per share have narrowed from $1.06 to 96 cents. CSTL shares have surged 38.3% over the past six months.
Castle Biosciences’ earnings beat estimates in three of the trailing four quarters and missed in the remaining one, with the average surprise being 66.11%.
Over the past 60 days, estimates for Recursion Pharmaceuticals’ 2026 loss per share have narrowed from $1.08 to $1.03. RXRX shares have risen 22.9% over the past six months.
Recursion Pharmaceuticals’ earnings beat estimates in one of the trailing four quarters and missed in the remaining three quarters, with the average negative surprise being 18.19%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
This article originally published on Zacks Investment Research (zacks.com).
| Jun-04 | |
| Jun-02 | |
| Jun-01 | |
| May-29 | |
| May-27 | |
| May-13 | |
| May-12 | |
| May-08 | |
| May-07 | |
| May-06 | |
| May-06 | |
| May-06 | |
| May-06 | |
| May-06 | |
| May-06 |
Join thousands of traders who make more informed decisions with our premium features. Real-time quotes, advanced visualizations, alerts, and much more.
Learn more about Finviz Elite