|
|
|

|
|||||

|
|
Catalyst Pharmaceuticals CPRX reported adjusted earnings of 68 cents per share for the fourth quarter of 2025, beating the Zacks Consensus Estimate of 42 cents. The company had recorded adjusted earnings of 70 cents in the year-ago quarter.
Total revenues, the majority of which comprised product revenues, amounted to $152.6 million in the reported quarter, representing growth of 8% year over year. The recorded figure also surpassed the Zacks Consensus Estimate of $140 million.
Catalyst Pharmaceuticals’ top line primarily comprised revenues from the sale of Firdapse, the first approved drug for the treatment of Lambert-Eaton myasthenic syndrome (LEMS) and the epilepsy drug Fycompa (perampanel) CIII. Revenues generated from the sale of the newly launched muscle disease drug, Agamree (vamorolone), also contributed to the top line.
Firdapse generated sales worth $97.6 million in the reported quarter, up 18% year over year, driven by organic sales growth. The reported figure beat the Zacks Consensus Estimate of $96.4 million as well as our estimate of $95.8 million. The drug has been witnessing strong demand, increasing prescription rates from LEMS patients and continued diagnosis of new LEMS patients.
In 2023, Catalyst Pharmaceuticals acquired exclusive rights to manufacture and supply Agamree from Santhera Pharmaceuticals through a licensing agreement. In late 2023, the FDA approved Agamree for treating Duchenne Muscular Dystrophy in patients aged two years and older, which gave the company a third approved product. The drug was commercially launched in the United States in the middle of March 2024.
In the reported quarter, Agamree generated revenues worth $35.3 million, up 68% year over year. The reported figure beat the Zacks Consensus Estimate of $31.3 million and our estimate of $33 million.
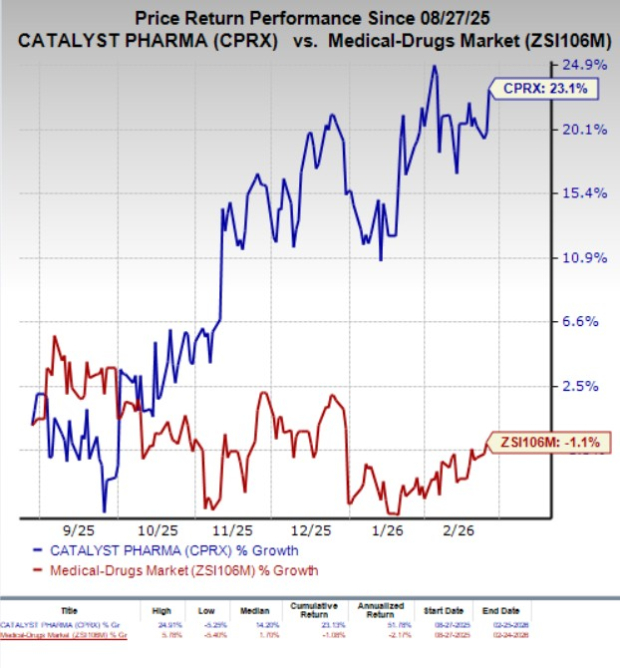
In the past six months, shares of CPRX have gained 23.1% against the industry’s 1.1% decline.
In 2023, Catalyst Pharmaceuticals acquired the U.S. rights for Fycompa (perampanel) CIII from Eisai Co., Ltd. This acquisition diversified the company’s portfolio by adding a commercial-stage epilepsy asset. Catalyst Pharmaceuticals started recording sales of Fycompa in 2023.
Fycompa generated net product revenues of $19.6 million, down significantly year over year, as tablet generics began hitting the market in May 2025 following the expiration of its first U.S. patent, with another slated to expire in July 2026. Catalyst Pharmaceuticals also lost exclusivity for the oral suspension version of Fycompa in December 2025. As a result, revenues from this product are expected to further decline in future periods as additional generic competition enters the market. The reported figure beat the Zacks Consensus Estimate of $12.3 million as well as our model estimate of $10.6 million.
Research and development expenses were $1.8 million in the reported quarter, down 52% year over year. Selling, general and administrative expenses totaled $53.4 million, up 21% year over year.
As of Dec. 31, 2025, Catalyst Pharmaceuticals had cash, cash equivalents and investments worth $709.2 million compared with $689.9 million as of Sept. 30, 2025.
In 2025, Catalyst Pharmaceuticals recorded total revenues of $589 million, up 20% year over year, beating the Zacks Consensus Estimate of $576.4 million, driven by higher product sales.
The company recorded adjusted earnings per share of $2.72 in 2025, which also surpassed the Zacks Consensus Estimate of $2.46. In 2024, CPRX recorded an earnings per share of $2.21.
In 2026, Catalyst Pharmaceuticals expects total revenues between $615 million and $645 million, reflecting continued growth in product revenues from Firdapse and Agamree.
Firdapse revenues are expected between $435 million and $450 million. Agamree revenues are anticipated between $140 million and $150 million in the current year. Per management, the guidance reflects its continued market adoption and commercial momentum.
Fycompa revenues are projected between $40 million and $45 million in 2026, driven by market demand despite the loss of patent exclusivity for Fycompa tablets, which currently has three generic competitors, and for Fycompa oral suspension, which currently has one generic competitor.
Catalyst Pharmaceuticals expects its R&D expenses to be between $17.5 million and $22.5 million in 2026. However, management stated that the expenses may shoot up in the event of an acquisition by the company. Although no numbers have been provided, CPRX expects its SG&A expenses to rise in 2025. The company further anticipates that its effective tax rate will be relatively consistent for 2026 compared with 2025 and 2024.

Catalyst Pharmaceuticals, Inc. price-consensus-eps-surprise-chart | Catalyst Pharmaceuticals, Inc. Quote
Catalyst Pharmaceuticals continues to evaluate Agamree in the SUMMIT study, a five-year follow-up study designed to evaluate the drug’s long-term clinical safety profile, including potential benefits on behavior, stature, bone health and cardiovascular health. The study is currently enrolling patients.
So far, CPRX has resolved Firdapse patent litigation with Teva Pharmaceuticals and Lupin Pharmaceuticals, with only one remaining case against Hetero USA. Trial is set to begin on March 23, 2026, ahead of the automatic 30-month stay expiration on May 26, 2026. The outcome remains uncertain.
Catalyst Pharmaceuticals currently carries a Zacks Rank #3 (Hold).
Some better-ranked stocks in the biotech sector are USANA Health Sciences USNA, Castle Biosciences CSTL and Recursion Pharmaceuticals RXRX. While USNA and CSTL sport a Zacks Rank #1 (Strong Buy) each, RXRX has a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.
Over the past 60 days, estimates for USANA Health Sciences’ 2026 earnings per share have risen from $1.90 to $2.00. USNA shares have lost 33.8% over the past six months.
USANA Health Sciences’ earnings beat estimates in three of the trailing four quarters and matched once, with the average surprise being 21.92%.
Over the past 60 days, estimates for Castle Biosciences’ 2026 loss per share have narrowed from $1.06 to 96 cents. CSTL shares have surged 38.3% over the past six months.
Castle Biosciences’ earnings beat estimates in three of the trailing four quarters and missed in the remaining one, with the average surprise being 66.11%.
Over the past 60 days, estimates for Recursion Pharmaceuticals’ 2026 loss per share have narrowed from $1.08 to $1.03. RXRX shares have risen 22.9% over the past six months.
Recursion Pharmaceuticals’ earnings beat estimates in one of the trailing four quarters and missed in the remaining three quarters, with the average negative surprise being 18.19%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
This article originally published on Zacks Investment Research (zacks.com).
| May-13 | |
| May-12 | |
| May-12 | |
| May-11 | |
| May-11 | |
| May-11 | |
| May-08 | |
| May-08 | |
| May-08 | |
| May-08 |
Angelini Pharma to acquire Catalyst Pharmaceuticals for equity value of $4.1bn
CPRX
Pharmaceutical Business Review
|
| May-07 | |
| May-07 | |
| May-07 | |
| May-07 | |
| May-06 |
Join thousands of traders who make more informed decisions with our premium features. Real-time quotes, advanced visualizations, alerts, and much more.
Learn more about Finviz Elite