|
|
|

|
|||||

|
|
GRI Bio, Inc.’s GRI shares plummeted 36.9% on April 1 after the company announced the pricing of a public offering of stock of $5 million.
Earlier in the day, the stock surged after the company reported positive interim safety results from its ongoing phase IIa study evaluating pipeline candidate GRI-0621 for the treatment of idiopathic pulmonary fibrosis (IPF).
Last month, the company received notice from The Nasdaq Stock Market LLC stating that it regained compliance with Nasdaq’s “minimum bid price rule.”
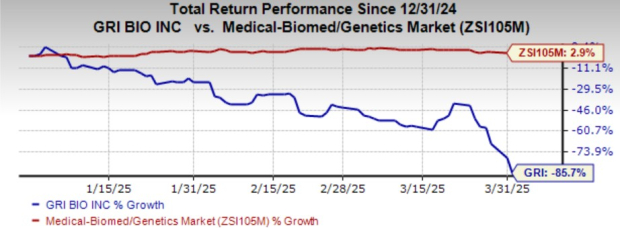
Shares of GRI have nosedived 85.7% year to date against the industry’s growth of 2.9%.
GRI Bio is offering 1.39 million shares of its common stock, Series E-1 warrants to purchase up to 1.39 million shares of common stock, short-term Series E-2 warrants to purchase up to 1.39 million shares of common stock and short-term Series E-3 warrants to purchase up to 1.39 million shares of common stock at a combined public offering price of $3.60 per share and accompanying warrants.
The warrants will have an exercise price of $3.20 per share. These will be exercisable immediately upon issuance. The closing of the offering is expected to occur on or about April 2, 2025, subject to the satisfaction of customary closing conditions.
In February, GRI Bio announced a reverse stock split (1-for-17) to increase the per share trading price of its common stock to regain compliance with the minimum bid price requirement for continued listing on The Nasdaq Capital Market.
The company’s lead product candidate, GRI-0621, is an oral inhibitor of type 1 invariant Natural Killer T (iNKT) cells. GRI-0621 is also an oral formulation of tazarotene, a synthetic RAR-beta and gamma selective agonist, that is approved in the United States for topical treatment of psoriasis and acne.
The pre-planned interim analysis for two-week safety results from the ongoing phase IIa biomarker study demonstrated GRI-0621 (4.5mg orally once daily) to be safe and well-tolerated in the first 12 patients evaluated per protocol.
The 12 patients assessed at the two-week visit did not show hyperlipidemia, as indicated by LDL, HDL and triglyceride (TG) levels. In addition, there were no meaningful changes in HDL, LDL or TG levels in patients receiving GRI-0621, and all subjects remained within normal ranges.
The Independent Data Monitoring Committee has recommended continuing the study as planned, as there are no safety concerns seen in the data reviewed.
The interim results show that GRI-0621’s receptor selectivity is consistent with the toxicity profile observed in earlier studies evaluating oral tazarotene in more than 1,700 patients treated for up to 52 weeks.
The phase IIa, randomized, double-blind, multi-center, placebo-controlled, parallel-design, 2-arm study will enroll approximately 36 subjects with IPF. These patients will be randomized in a 2:1 ratio to receive either GRI-0621 4.5mg or a placebo.
A sub-study will examine the number and activity of NKT cells in bronchoalveolar lavage (BAL) fluid for up to 12 eligible subjects (across various centers).
An interim analysis will be performed when 24 subjects (of which approximately eight will be placebo subjects) complete six weeks of treatment.
GRI Bio expects to report interim biomarker data in the second quarter of 2025. Top-line results from the phase IIa biomarker study are expected in the third quarter of 2025.
Positive results from the study will be a boost for GRI stock, which is currently sailing in troubled waters.
GRI Bio obtained two global patents, thereby strengthening its intellectual property portfolio. The patent claims include coverage of GRI-0803, GRI’s novel activator of human type 2 diverse NKT (dNKT) cells in development for the treatment of autoimmune disorders, with an initial focus on systemic lupus erythematosus.
The other patent covers claims including compositions and methods for modulating dNKT and/or iNKT cells in the prevention and treatment of inflammatory conditions.
GRI Bio currently carries a Zacks Rank #3 (Hold). Some better-ranked stocks in the biotech sector are Amicus Therapeutics, Inc. FOLD and ANI Pharmaceuticals, Inc. ANIP, both carrying a Zacks Rank #1 (Strong Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.
In the past 60 days, estimates for Amicus’ earnings per share (EPS) have increased from 42 to 52 cents for 2025. During the same time frame, EPS estimates for 2026 have increased from 70 to 78 cents.
In the past 60 days, estimates for ANI Pharmaceuticals’ EPS have increased from $5.54 to $6.35 for 2025. During the same period, EPS estimates for 2026 have increased from $6.75 to $7.21. Year to date, shares of ANIP have rallied 23%.
ANIP’s earnings beat estimates in each of the trailing four quarters, the average surprise being 17.32%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
This article originally published on Zacks Investment Research (zacks.com).
| May-27 | |
| May-18 | |
| May-14 | |
| May-09 | |
| May-08 | |
| May-08 | |
| May-08 | |
| Apr-28 |
BioMarin acquires Amicus Therapeutics for $4.8bn
Pharmaceutical Business Review
|
| Apr-24 | |
| Apr-20 | |
| Apr-13 | |
| Apr-08 | |
| Apr-08 | |
| Apr-08 | |
| Apr-07 |
Join thousands of traders who make more informed decisions with our premium features. Real-time quotes, advanced visualizations, alerts, and much more.
Learn more about Finviz Elite