|
|
|

|
|||||

|
|
Eli Lilly’s LLY key top-line drivers are its GLP-1 medicines, Mounjaro for type II diabetes and Zepbound for obesity. Despite being on the market for slightly more than three years, Mounjaro and Zepbound have seen exceptional sales growth, with demand rising rapidly. These therapies account for more than 50% of the company’s total revenues.
Mounjaro and Zepbound include the same compound, tirzepatide, a dual GIP and GLP-1 receptor agonist. The GLP-1 segment is a very important class of drugs for multiple cardiometabolic diseases and has gained significant popularity.
Mounjaro is the market leader in new prescriptions within type II diabetes incretin analogs in both the United States and ex-U.S. markets, while Zepbound also holds a leading market share in the branded obesity market with nearly 70% share of new prescriptions.
Mounjaro and Zepbound face strong competition from Novo Nordisk’s NVO semaglutide medicines, Ozempic for diabetes and Wegovy for obesity. However, Lilly’s Mounjaro and Zepbound have generated stronger sales and captured greater market share than Ozempic and Zepbound.
In 2025, the drugs generated combined sales of $36.5 billion, comprising around 56% of the company’s total revenues. Robust growth trends in the U.S. incretin analogs market and positive uptake trends in new international markets led to strong sales growth in 2025. The trend is expected to continue in 2026.
Approvals for new obesity related conditions can also drive sales of Mounjaro and Zepbound higher. In 2025, Lilly launched additional Zepbound lower-priced vial doses, which are showing sustained growth uptake and offered new savings for self-pay patients. Lilly is also expanding its incretin production capability. All these efforts can help sustain Mounjaro and Zepbound’s growth in future quarters amid rising competitive pressure.
The launch of Lilly’s oral GLP-1 pill called orforglipron is expected to expand the addressable market. Lilly has already filed regulatory applications in the United States, the EU and several other countries seeking approval for orforglipron in obesity. Lilly expects to launch orforglipron for obesity in the United States in the second quarter of 2026 and in most international markets during 2027.
Novo Nordisk gained approval for an oral version of Wegovy in December 2025 and launched the pill in January 2026.
In addition, Lilly expects new Medicare access to obesity medicines to become effective, no later than July 2026, which should accelerate adoption. While Medicaid access to obesity drugs is expected to decrease in 2026 due to key states like California removing obesity coverage, Lilly is hoping that new states will add coverage for people with Medicaid in 2027.
With Mounjaro and Zepbound anchoring more than half of total revenues and new catalysts on the horizon — including expanded production, broader Medicare access and the upcoming launch of oral orforglipron — Lilly is doubling down on its GLP-1 leadership as competition in the fast-growing obesity market intensifies.
The global obesity drug market is projected to grow dramatically, reaching nearly $95 billion by 2030 and potentially $125 billion by 2035, according to Goldman Sachs estimates. While Lilly and Novo Nordisk currently dominate this space, both companies are racing to develop next-generation, more powerful and more convenient GLP-1–based treatments, including oral options and multi-acting candidates, to stay ahead of emerging rivals.
Smaller biotechs like Structure Therapeutics GPCR and Viking Therapeutics VKTX are also developing oral GLP-1 drugs for treating obesity.
Viking Therapeutics’ dual GIPR/GLP-1 receptor agonist, VK2735, is being developed both as oral and subcutaneous formulations for the treatment of obesity. Viking plans to advance oral VK2735 into phase III development for obesity in the third quarter of 2026.
Structure Therapeutics’ ACCESS study on its orally GLP-1 RA, aleniglipron, for obesity, met its primary and all key secondary endpoints. Structure Therapeutics expects to initiate the late-stage program of aleniglipron in obesity around mid-2026.
Others like Roche, Merck and AbbVie are also looking to enter the obesity space by in-licensing obesity candidates from smaller biotechs, which could threaten Novo Nordisk and Eli Lilly’s dominance in the market. In November, Pfizer acquired obesity drugmaker Metsera to gain a foothold in the space.
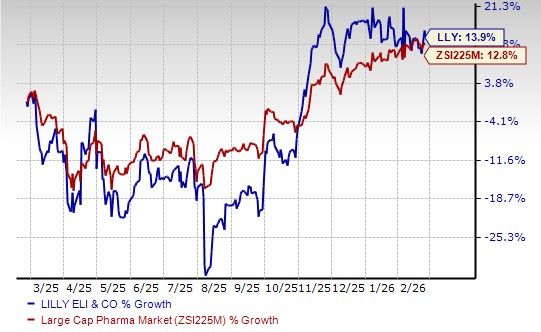
Lilly’s stock has risen 13.9% in the past year compared with the industry’s increase of 12.8%.
From a valuation standpoint, Lilly’s stock is expensive. Going by the price/earnings ratio, the company’s shares currently trade at 29.69 forward earnings, higher than 18.69 for the industry. However, the stock is trading below its 5-year mean of 34.57.

The Zacks Consensus Estimate for 2026 has risen from $33.14 to $33.86 per share over the past 30 days, while that for 2027 has risen from $41.48 to $41.96 per share over the same timeframe.

Lilly has a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
This article originally published on Zacks Investment Research (zacks.com).
| 5 hours | |
| 7 hours | |
| 9 hours | |
| Jun-09 | |
| Jun-09 | |
| Jun-09 | |
| Jun-09 | |
| Jun-09 |
Dow Jones AI Giant Nvidia Stock Tumbles To Key Support Level Amid Market Sell-Off
LLY
Investor's Business Daily
|
| Jun-09 | |
| Jun-09 | |
| Jun-09 | |
| Jun-09 | |
| Jun-09 | |
| Jun-09 | |
| Jun-09 |
Join thousands of traders who make more informed decisions with our premium features. Real-time quotes, advanced visualizations, alerts, and much more.
Learn more about Finviz Elite